Molecular Formula
C15H13NO3S2
Smiles
C/C(=C\C1=CC=CC=C1)/C=C\2/C(=O)N(C(=S)S2)CC(=O)O
General Description
Epalrestat is a specific, oral, non-competitive and reversible aldose reductase (AR) inhibitor. AR is the enzyme that catalyzes the first rate-limiting step of the polyol pathway. Epalrestat has a broad cellular protective profile against multiple pathways of metabolic stress that cause cellular damage, and it has antioxidant and anti-inflammatory properties as well as putative antineoplastic activity.
Mechanism of Action
Epalrestat exerts its protective effect by inhibiting the conversion of glucose to sorbitol, thereby preventing intracellular polyol accumulation and re-establishing myo-inositol content and Na+-K+-ATPase activity. Epalrestat's inhibition of AR activity diminishes the production of reactive oxygen species (ROS) and advanced glycation end-products (AGEs). Additionally, it modulates the activity of the NF-kappa B and Protein Kinase C (PKC) signaling pathways, which are involved in the regulation of inflammatory cytokine expression and oxidative stress responses.
Application
Epalrestat is mainly used for the treatment and amelioration of symptoms associated with diabetic complications, especially the symptoms of diabetic peripheral neuropathy (pain, numbness, abnormal vibration sense). It is also used for autonomic nervous system dysfunction, retinal microvascular lesion, and corneal microvascular lesion.
Epalrestat is the only aldose-reductase inhibitor approved for diabetic neuropathy in Japan. Using rat Schwann cells, the authors demonstrate that clinically relevant concentrations of Epalrestat 3-5 fold increase intracellular glutathione by transcriptionally up-regulating the rate-limiting enzyme in GSH synthesis, γ-glutamylcysteine synthetase (γ-GCS). This effect is mediated by nuclear translocation and activation of Nrf2, as shown by ELISA and is abolished by Nrf2 siRNA. The elevated GSH confers striking resistance to oxidative insults.
The results identify Epalrestat as an Nrf2 activator that boosts endogenous antioxidant defenses and thus suggest a new clinical utility for this drug beyond glycaemic control for oxidative stress-related disorders such as neuropathy, atherosclerosis and neurodegeneration.
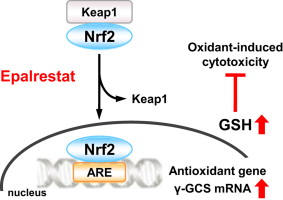 Fig. 1 Epalrestat increases intracellular glutathione levels. (Sato K.; et al. 2014)
Fig. 1 Epalrestat increases intracellular glutathione levels. (Sato K.; et al. 2014)
References
- Sato K, et al. Epalrestat increases intracellular glutathione levels in Schwann cells through transcription regulation. Redox biology, 2014, 2: 15-21.
Epalrestat, an aldose reductase inhibitor with poor water solubility, was combined with β-cyclodextrin or sulfobutyl ether β-CD (SBE7β-CD) to form inclusion complexes, which were then encapsulated into chitosan (CS) nanoparticles via ionotropic gelation. Six EPL-CD-loaded CS nanoparticle formulations were prepared and optimized. NP6 (EPL-SBE7β-CD, coprecipitation method) showed the smallest size, highest drug entrapment efficiency, and fastest drug release. FTIR, PXRD, DSC, SEM, and TEM confirmed successful complexation, amorphous drug state, and spherical morphology. In-silico docking supported stable interaction between SBE7β-CD and the cross-linker TPP.
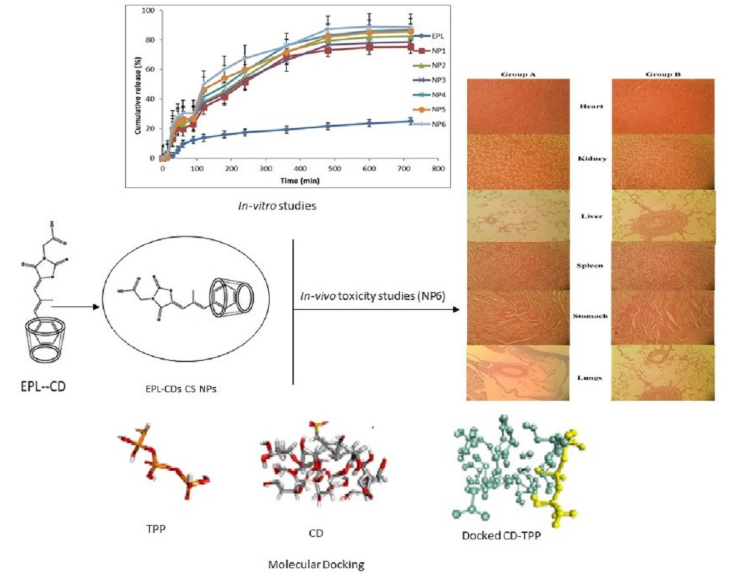 Fig. 2 Nanoparticles loaded with Epalrestat-cyclodextrin inclusion complex. (Alvi Z.; et al. 2021)
Fig. 2 Nanoparticles loaded with Epalrestat-cyclodextrin inclusion complex. (Alvi Z.; et al. 2021)
References
- Alvi Z, et al. Utilization of gelling polymer to formulate nanoparticles loaded with epalrestat-cyclodextrin inclusion complex: formulation, characterization, in-silico modelling and in-vivo toxicity evaluation. Polymers, 2021, 13(24): 4350.
What are the major uses of Epalrestat?
It is used for the treatment and management of diabetic peripheral neuropathy symptoms.
Does your supply of Epalrestat meet the criteria for research and development?
Yes, we offer high-purity Epalrestat for pharmaceutical R&D and formulation studies.
Do you supply Epalrestat in bulk?
Yes, we can supply Epalrestat in bulk for commercial production.
Is Epalrestat a competitive or non-competitive inhibitor?
Epalrestat is a non-competitive, reversible inhibitor of aldose reductase.


 Fig. 1 Epalrestat increases intracellular glutathione levels. (Sato K.; et al. 2014)
Fig. 1 Epalrestat increases intracellular glutathione levels. (Sato K.; et al. 2014)
 Fig. 2 Nanoparticles loaded with Epalrestat-cyclodextrin inclusion complex. (Alvi Z.; et al. 2021)
Fig. 2 Nanoparticles loaded with Epalrestat-cyclodextrin inclusion complex. (Alvi Z.; et al. 2021)













