Storage
Store at <-15°C, close container well
Synonyms
TAK-438; 1-(5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine Fumarate; 5-(2-Fluorophenyl)-N-methyl-1-(3-pyridinylsulfonyl)-1H-pyrrole-3-methanamine (2E)-2-butenedioate (1:1); Takecab
Molecular Formula
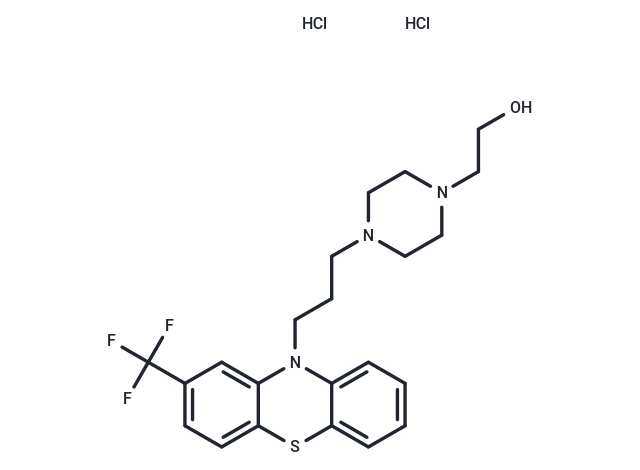
C21H20FN3O6S
Smiles
CNCC1CN(C(C1)C1CCCCC1F)S(=O)(=O)C1CNCCC1.C(=C\C(=O)O)/C(=O)O
General Description
Vonorazan fumarate is the fumarate salt of vonorazan. Vonorazan's chemical structure is different from other P-CABs (such as SCH 28080 and AZD0865 that have a benzimidazole ring) or revivalazine (that has a pyrimidine ring). It is a pyrrole derivative and is part of the class of reversible potassium-competitive acid blockers (P-CABs), and has potential anti-acid activity.
Mechanism of Action
Vonoprazan fumarate is a novel first-in-class potassium-competitive acid blocker (P-CAB), which introduces a new mechanism for gastric acid suppression. It differs from currently available proton pump inhibitors (PPIs), which are prodrugs requiring acid activation and binding irreversibly to the proton pump. Vonoprazan directly inhibits the H+, K+-ATPase enzyme in a reversible and potassium-competitive manner. It does not require acid activation due to high pKa, and as such results in rapid, potent and sustained acid inhibition even at the first dose.
Application
Indications for vonoprazan fumarate mainly include the treatment of acid-related diseases such as erosive esophagitis, gastric ulcer, and duodenal ulcer. Its role in Helicobacter pylori eradication therapy has gained a lot of importance recently, as the better acid suppression with vonoprazan leads to a more stable environment for the antibiotics that are administered with it. Vonoprazan fumarate is also used to prevent recurrent ulcers in patients taking NSAIDs.
Vonoprazan is new potential gastric acid suppression agent, and it is classified as potassium-competitive acid blocker (P-CAB). Potassium-competitive acid blocker, which is different from PPI, enters parietal cell canaliculus in an active form, is stable in an acidic environment, and does not require acid activation. Protonated P-CAB will make non-covalent bonds with H+/K+-ATPase, thus inactivating the H+/K+-ATPase with a slower dissociation rate and for a longer time.
 Fig. 1 The mode of action of proton pump inhibitors (PPIs) and Vonoprazan against H. pylori infection. (Miftahussurur M, et al. 2020)
Fig. 1 The mode of action of proton pump inhibitors (PPIs) and Vonoprazan against H. pylori infection. (Miftahussurur M, et al. 2020)
References
- Miftahussurur M, et al. The potential benefits of vonoprazan as Helicobacter pylori infection therapy. Pharmaceuticals. 2020, 13(10): 276.
Vonoprazan fumarate (VPF) is a novel potassium-competitive acid blocker with potential applications in the management of acid-related diseases and Helicobacter pylori (H. pylori) eradication. However, the poor solubility and low bioavailability of VPF impede its therapeutic efficacy. To address these challenges, researchers developed amorphous solid dispersion (ASD)-loaded orodispersible films (ODFs). VPF ASDs were prepared with hydroxypropyl methylcellulose (HPMC K4M), polyethylene glycol 4000, and Poloxamer 407 using a melt-based approach. The optimized VPF ASD, showing the best dissolution, was formulated at a 1: 6 drug-to-polymer ratio with HPMC K4M and followed the Higuchi-diffusion kinetic model. The ASD was subsequently loaded into ODFs prepared by solvent casting using different polymers, and ODF7 (75 mg polyvinyl alcohol) exhibited the fastest disintegration, least moisture uptake, and 100% drug release in 10 minutes. The in vivo pharmacokinetic studies in rabbits showed a 2.2-fold increase in relative bioavailability (221.98%) compared to commercial tablets and palatability. These results support the use of VPF-ASD-loaded ODFs as a suitable approach for the enhancement of solubility, stability, and systemic exposure, as well as patient compliance.
 Fig. 2 SEM of pure VPF and VPF-HPMC-ASD. (El-Shenawy A A, Abd Elkarim R A. 2026)
Fig. 2 SEM of pure VPF and VPF-HPMC-ASD. (El-Shenawy A A, Abd Elkarim R A. 2026)
References
- El-Shenawy A A, Abd Elkarim R A. Fabrication and in-vitro/in-vivo characterization of orodispersible films loaded with amorphous solid dispersion of vonoprazan fumarate. Journal of Pharmaceutical Innovation. 2026, 21(1): 71.
Does Vonoprazan Fumarate require protection from light during storage?
Yes, store Vonoprazan Fumarate in light-resistant containers to maintain chemical stability.
Can Vonoprazan Fumarate be stored near acidic materials?
No, keep Vonoprazan Fumarate separate from reactive acids to prevent degradation.
How can I track my Vonoprazan Fumarate shipment online?
We provide a tracking number immediately after the Vonoprazan Fumarate leaves our facility.
Can I modify my Vonoprazan Fumarate order after placement?
Modifications are possible before the Vonoprazan Fumarate enters the shipping stage.


 Fig. 1 The mode of action of proton pump inhibitors (PPIs) and Vonoprazan against H. pylori infection. (Miftahussurur M, et al. 2020)
Fig. 1 The mode of action of proton pump inhibitors (PPIs) and Vonoprazan against H. pylori infection. (Miftahussurur M, et al. 2020)
 Fig. 2 SEM of pure VPF and VPF-HPMC-ASD. (El-Shenawy A A, Abd Elkarim R A. 2026)
Fig. 2 SEM of pure VPF and VPF-HPMC-ASD. (El-Shenawy A A, Abd Elkarim R A. 2026)