Storage
Keep in dark place, sealed in dry, 2-8°C
Synonyms
NCT; 4-Nitrophenyl (thiazol-5-ylmethyl) carbonate; 4-Nitrophenyl(thiazol-5-ylmethyl)carbonate; Thiazolylmethyl-4-nitrophenylcarbonate; SY102959; ((5-Thiazolyl)methyl)-(4-nitrophenyl)carbonate
Molecular Formula
C11H8N2O5S
Smiles
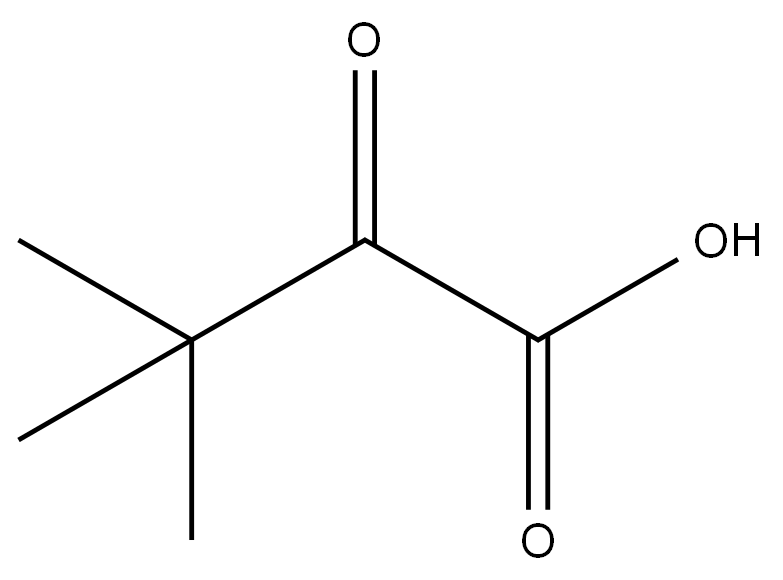
O=C(OCC1=CN=CS1)OC2=CC=C([N+]([O-])=O)C=C2
Boiling Point
475.333ºC at 760 mmHg
General Description
4-Nitrophenyl 1,3-thiazol-5-ylmethyl carbonate (NCT) is one of the most central chiral intermediates in the synthesis of the antiviral drug ritonavir. It forms the "backbone" of the Ritonavir molecular structure and contains a critical hydroxyethylamine isostere. This specific structure mimics the transition state of a protein substrate, which enables the final drug to effectively inhibit viral protease activity. The production of NCT involves complex multi-step chiral synthesis processes, requiring exceptionally high standards for both optical purity (EE value) and chemical purity.
Application
4-Nitrophenyl 1,3-thiazol-5-ylmethyl carbonate (NCT) is primarily utilized in the industrial-scale production of high-performance antiretroviral medications, serving as the essential raw material for the preparation of Ritonavir. Within the synthetic pathway, NCT undergoes coupling reactions with specific isocyanates or amino acid derivatives, eventually undergoing condensation to form the active pharmaceutical ingredient (API)-ritonavir.
Ritonavir irreversibly inactivates CYP3A4/5 enzymes by four mechanisms that "boost" other drugs by preventing their metabolism.
Metabolic Intermediate Complex (MIC): A ritonavir metabolite ('believed to be the primary amine M1') binds tightly to the ferrous (+2) oxidation state of the heme iron and serves as an irreversible, non-covalent trap for binding in the active site.
Tight Binding to Heme Iron: Binding of unchanged ritonavir directly to the heme iron. Ritonavir serves as a Type II ligand and its thiazole nitrogen binds tightly to the heme iron. The coordination of ritonavir to the heme iron lowers the redox potential of the iron so much that it becomes thermodynamically unfavorable for electrons from CPR (the enzyme's redox partner) to reduce it for catalysis to occur.
Heme Destruction: The enzyme metabolizes ritonavir to a reactive intermediate that forces the destruction of heme and subsequent covalent linkage to the apoprotein.
Covalent Binding to Apoprotein: Ritonavir metabolizes into a reactive intermediate (possibly a sulfoxide) that covalently binds to lysine residue Lys257 of the enzyme's CYP3A4 polypeptide, effectively "locking" the protein in place.
 Fig. 1 An overview of the four main mechanisms of inactivation of CYP3A4/5 by ritonavir. (Loos N H C, et al. 2022)
Fig. 1 An overview of the four main mechanisms of inactivation of CYP3A4/5 by ritonavir. (Loos N H C, et al. 2022)
References
- Loos N H C, et al. The mechanism-based inactivation of CYP3A4 by ritonavir: what mechanism. International Journal of Molecular Sciences. 2022, 23(17): 9866.
Ritonavir (RN), an anti-retroviral protease inhibitor used against HIV/AIDS, was prepared in solid lipid nanoparticles (RN-SLNs) using a hot homogenization-ultrasonication technique optimized by employing central composite design having phospholipids and surfactants as independent variables. The highest solubility of RN was exhibited by soya lecithin (103.34 mg/g), while the highest entrapment efficiency was observed with Tween 80:Poloxamer 188 (8:2) as surfactant mixture. The optimized RN-SLNs exhibited a particle size of 265.06 nm, 86.2% entrapment efficiency, and 94.8% cumulative drug release, following Higuchi’s model with biphasic release. In vivo studies revealed a 4.3-fold increase in bioavailability compared to marketed formulations, providing a promising technological platform for improving the therapeutic delivery of anti-retroviral treatments.
 Fig. 2 Enhanced oral bioavailability of ritonavir loaded SLLN. (Chettupalli A K, et al. 2025)
Fig. 2 Enhanced oral bioavailability of ritonavir loaded SLLN. (Chettupalli A K, et al. 2025)
References
- Chettupalli A K, et al. Ritonavir loaded solid lipid nanoparticles for oral drug delivery and bioavailability enhancement. Discover Applied Sciences. 2025, 7(1): 58.
Should 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate be protected from humidity?
Yes, keep 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate in a tightly sealed container to avoid moisture.
Can 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate be stored near oxidizing agents?
No, keep 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate separate from strong oxidizers to avoid reactions.
What documents accompany the 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate shipment?
Each box includes the 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate COA, invoice, and packing list.
How can I obtain a 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate price list?
Submit an inquiry form to receive current 4-Nitrophenyl 1,3-Thiazol-5-Ylmethyl Carbonate market pricing.


 Fig. 1 An overview of the four main mechanisms of inactivation of CYP3A4/5 by ritonavir. (Loos N H C, et al. 2022)
Fig. 1 An overview of the four main mechanisms of inactivation of CYP3A4/5 by ritonavir. (Loos N H C, et al. 2022)
 Fig. 2 Enhanced oral bioavailability of ritonavir loaded SLLN. (Chettupalli A K, et al. 2025)
Fig. 2 Enhanced oral bioavailability of ritonavir loaded SLLN. (Chettupalli A K, et al. 2025)


![tert-butyl 3,8-diazaspiro[5.6]dodecane-3-carboxylate](https://www.protheragen.ai/wp-content/themes/protheragen-ing/img/StructureImage58.gif)









