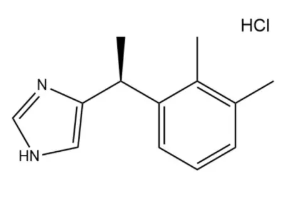
Synonyms
8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine hydrochloride
Molecular Formula
C18H14Cl2FN3
Smiles
CC1=NC=C2N1C3=C(C=C(C=C3)Cl)C(=NC2)C4=CC=CC=C4F.Cl
Appearance
White to light yellow crystalline compound
General Description
Midazolam hydrochloride is the water-soluble salt form of midazolam, a short-acting benzodiazepine central nervous system depressant. At physiologic pH, the water-soluble hydrochloride salt converts to the lipophilic free base, enabling rapid penetration of the blood-brain barrier.
Mechanism of Action
Midazolam produces its effects by binding to the benzodiazepine receptor at the gamma-aminobutyric acid (GABA) receptor-chloride ionophore complex in the central nervous system. This binding enhances the affinity of GABA for its receptor, increasing the frequency of chloride channel opening and resulting in membrane hyperpolarization. The pharmacodynamic consequences include anxiolysis, sedation, amnesia, anticonvulsant activity, and muscle relaxation directly proportional to receptor occupancy.
Application
Midazolam hydrochloride is indicated for conscious sedation prior to diagnostic or therapeutic procedures, anesthesia premedication, induction of anesthesia, and as a sedative component in combined anesthesia. It is also used for continuous sedation in intensive care units and for treatment of status epilepticus.
Jiao J, et al. summarized the molecular mechanisms through which midazolam may exert anticancer effects, including inhibition of cell proliferation, induction of apoptosis, and suppression of migration and invasion in various cancer cell lines. However, the precise pathways remain to be fully elucidated, and the evidence is primarily preclinical. The authors conclude that while midazolam shows promise as a potential therapeutic agent in oncology, further studies are needed to clarify its mechanisms of action and to determine its clinical relevance and safety in cancer patients.
 Fig. 1 Flow diagram of the studies identification and selection. (Jiao J, et al., 2017)
Fig. 1 Flow diagram of the studies identification and selection. (Jiao J, et al., 2017)
References
- Jiao J, et al. Insights into the Roles of Midazolam in Cancer Therapy. Evid Based Complement Alternat Med. 2017;2017:3826506.
Gershuny V, et al. reported a randomized, double-blind, four-period crossover trial involving 20 healthy volunteers to investigate whether midazolam co-administered with oxycodone exacerbates respiratory depression compared to oxycodone alone. Using modified Read rebreathing methodology, the combination did not significantly decrease minute ventilation at elevated end-tidal CO₂ (7.3 kPa) compared to oxycodone alone. However, midazolam plus oxycodone increased resting end-tidal CO₂ and caused 60% of participants to fall asleep or snore compared to 0% with oxycodone alone.
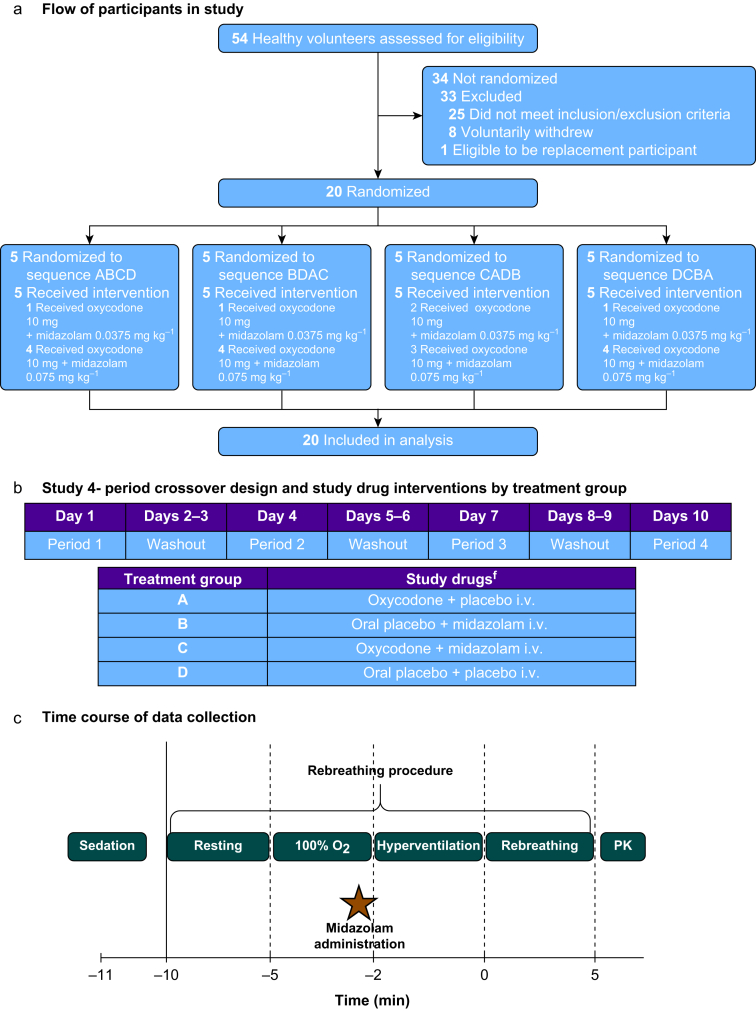 Fig. 2 Flow of participants in the study, interventions, and overall study design. (Gershuny V, et al, 2025)
Fig. 2 Flow of participants in the study, interventions, and overall study design. (Gershuny V, et al, 2025)
References
- Gershuny V, et al. Effect of midazolam co-administered with oxycodone on ventilation: a randomised clinical trial in healthy volunteers. Br J Anaesth. 2025;134(4):1170-1180.
Does Midazolam Hydrochloride require strict temperature control during air freight?
Yes, it should be maintained at 15-25°C during air freight. We use validated thermal shippers with data loggers to ensure no deviation occurs during transit.
Is Midazolam Hydrochloride prone to hydrolysis in the presence of moisture?
The hydrochloride salt is more stable than the free base, but it is still advisable to protect it from excessive humidity to prevent caking and potential ring-opening hydrolysis.
What documentation is required for international shipping of Midazolam as a controlled substance?
We provide all necessary export permits, certificates of origin, and controlled substance licenses to ensure smooth customs clearance in compliance with international regulations.
How is the impurity profile of Midazolam Hydrochloride monitored over time?
Stability studies specifically track the formation of the benzophenone derivative, a primary degradation product, using a validated, stability-indicating HPLC assay.


 Fig. 1 Flow diagram of the studies identification and selection. (Jiao J, et al., 2017)
Fig. 1 Flow diagram of the studies identification and selection. (Jiao J, et al., 2017)
 Fig. 2 Flow of participants in the study, interventions, and overall study design. (Gershuny V, et al, 2025)
Fig. 2 Flow of participants in the study, interventions, and overall study design. (Gershuny V, et al, 2025)