Synonyms
HCTZ; Esidrix; Hydro-Diuril; Oretic
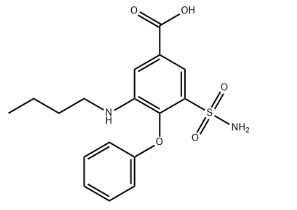
Molecular Formula
C7H8ClN3O4S2
Smiles
C1NC2=CC(=C(C=C2S(=O)(=O)N1)S(=O)(=O)N)Cl
General Description
Hydrochlorothiazide is a thiazide-type diuretic and antihypertensive agent widely used for over 60 years. As the most frequently prescribed thiazide diuretic, it is characterized by its reliable efficacy and relatively safe profile for managing hypertension and edema.
Mechanism of Action
Hydrochlorothiazide exerts its effects by directly inhibiting the sodium chloride cotransporter located on the apical membrane of the distal convoluted tubules in the kidney. This inhibition prevents sodium reabsorption, inducing natriuresis and diuresis that initially lower blood pressure through volume loss. Over time, it sustains blood pressure reduction through vasodilation and decreased peripheral vascular resistance, though the exact mechanism for this effect remains poorly understood.
Application
Hydrochlorothiazide is FDA-approved for treating essential hypertension as primary or adjunctive therapy and for managing peripheral edema associated with heart failure, cirrhosis, corticosteroids, or nephrotic syndrome.
Vasco RFV, et al. compared the effects of furosemide and hydrochlorothiazide on parameters of chronic kidney disease–mineral and bone disorder (CKD-MBD) in patients with stage 3 CKD followed for one year. Furosemide treatment resulted in increased bone remodeling and elevated parathyroid hormone (PTH) levels, suggesting stimulation of bone turnover. In contrast, hydrochlorothiazide attenuated the rise in PTH and decreased bone turnover markers. These divergent effects reflect the distinct mechanisms of action of loop versus thiazide diuretics on calcium handling and mineral metabolism.
 Fig. 1 Effect of diuretic type on PTH 1-84 and bone biomarkers. (Vasco RFV, et al., 2021)
Fig. 1 Effect of diuretic type on PTH 1-84 and bone biomarkers. (Vasco RFV, et al., 2021)
References
- Vasco RFV, et al. Effects of diuretics furosemide and hydrochlorothiazide on CKD-MBD: A prospective randomized study. Bone Rep. 2021;14:100746.
Vaculikova E, et al. conducted a pharmaceutical development study aimed at improving the solubility of hydrochlorothiazide, a Class IV Biopharmaceutical Classification System drug, through nanoparticle formulation. Using antisolvent precipitation–solvent evaporation and emulsion solvent evaporation methods with various surfactants, the researchers successfully prepared 18 nanoparticle samples. The optimal formulation, stabilized with 5% carboxymethyl dextran sodium salt, achieved a particle size of 2.6 nm and demonstrated 6.5-fold higher solubility compared to bulk hydrochlorothiazide.
 Fig. 2 SEM images of bulk hydrochlorothiazide at magnification 1600× (A) and hydrochlorothiazide stabilized with 5% carboxymethyl dextran sodium salt at magnification 100× (B). (Vaculikova E, et al, 2016)
Fig. 2 SEM images of bulk hydrochlorothiazide at magnification 1600× (A) and hydrochlorothiazide stabilized with 5% carboxymethyl dextran sodium salt at magnification 100× (B). (Vaculikova E, et al, 2016)
References
- Vaculikova E, et al. Preparation of Hydrochlorothiazide Nanoparticles for Solubility Enhancement. Molecules. 2016;21(8):1005.
Does Hydrochlorothiazide undergo photodegradation during storage?
Yes, it is sensitive to light, particularly UV radiation, which can lead to the formation of degradation products. It should be stored in light-resistant containers.
What is the recommended long-term storage temperature for Hydrochlorothiazide?
Controlled room temperature (20-25°C) is ideal. It is stable under normal conditions but should be protected from excessive heat and humidity to maintain its crystalline form.
Is Hydrochlorothiazide susceptible to alkaline hydrolysis?
In solution, it can hydrolyze at alkaline pH. For bulk API storage, the solid form is stable.
How is the impurity 4-amino-6-chloro-1,3-benzenedisulfonamide controlled?
This key related substance is specifically monitored using a validated HPLC method to ensure it remains below pharmacopoeial limits throughout the shelf life.


 Fig. 1 Effect of diuretic type on PTH 1-84 and bone biomarkers. (Vasco RFV, et al., 2021)
Fig. 1 Effect of diuretic type on PTH 1-84 and bone biomarkers. (Vasco RFV, et al., 2021)
 Fig. 2 SEM images of bulk hydrochlorothiazide at magnification 1600× (A) and hydrochlorothiazide stabilized with 5% carboxymethyl dextran sodium salt at magnification 100× (B). (Vaculikova E, et al, 2016)
Fig. 2 SEM images of bulk hydrochlorothiazide at magnification 1600× (A) and hydrochlorothiazide stabilized with 5% carboxymethyl dextran sodium salt at magnification 100× (B). (Vaculikova E, et al, 2016)