Storage
Store at 10°C - 25°C, close container well, protect from light.
Synonyms
(S)-Omeprazole; (-)-Omeprazole; 5-Methoxy-2-((S)-((4-methoxy-3,5-dimethyl-2-pyridyl)methyl)sulfinyl)benzimidazole; 6-methoxy-2-[(S)-(4-methoxy-3,5-dimethylpyridin-2-yl)methylsulfinyl]-1H-benzimidazole
Molecular Formula
C17H19N3O3S
Smiles
CC1=CN=C(C(=C1OC)C)C[S@](=O)C2=NC3=C(N2)C=C(C=C3)OC
Boiling Point
600ºC at 760mmHg
General Description
Esomeprazole is the S-configuration enantiomer of omeprazole, defined by its 5-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole structure. As a proton pump inhibitor, it effectively suppresses gastric acid secretion.
Mechanism of Action
Esomeprazole suppresses stomach acid by covalently binding to the sulfhydryl groups of cysteines on the (H+, K+)-ATPase enzyme located on the secretory surface of gastric parietal cells and thereby inhibiting the final step in the production of gastric acid. Esomeprazole inhibits both basal and stimulated gastric acid secretion regardless of the stimulus. The binding of esomeprazole to (H+, K+)-ATPase enzyme is irreversible and the new enzyme needs to be expressed before gastric acid secretion can resume.
Application
Applications for esomeprazole include the management of gastroesophageal reflux disease (GERD), gastric protection to prevent the recurrence of stomach ulcers or gastric damage from chronic NSAID use, and treatment of pathological hypersecretory conditions, such as Zollinger-Ellison (ZE) Syndrome. Esomeprazole is also used in quadruple regimens to treat H. pylori with other antibiotics.
Proton pump inhibitors (PPIs) act on the final step of gastric acid secretion by irreversibly blocking H⁺–K⁺-ATPase in parietal cells. The proton pump works to achieve a million-fold H⁺ gradient by pumping cytoplasmic H₃O⁺ out of the cell into the lumen in exchange for K⁺. The enzyme is stimulated by ligands (histamine/gastrin) to move from an inactive form on tubulovesicles to an active form in the secretory canaliculus. When it becomes activated, the enzyme undergoes conformational changes between the E1 state, in which the H₃O⁺ is bound on the cytoplasmic side, and E2, in which K⁺ is bound on the luminal side. Continuous activity of the proton pump requires recycling of K⁺ back into the lumen through the acid-sensitive KCNQ1/KCNE2 channel, which is open at pH 1. Secretion of HCO₃⁻ is necessary to preserve electroneutrality. PPIs take advantage of this transport system because they are acid-activated prodrugs that convert into sulfenamide derivatives. The reactive sulfenamides link to cysteine residues of the pump's α-subunit by the formation of disulfide bridges. The ion transport and conformational changes are now blocked for the lifetime of the enzyme, and acid secretion is inhibited until new pumps are produced.
 Fig. 1 General chemical structure and mechanism of action of proton pump inhibitors (PPIs). (Ward R M, Kearns G L. 2013)
Fig. 1 General chemical structure and mechanism of action of proton pump inhibitors (PPIs). (Ward R M, Kearns G L. 2013)
References
- Ward R M, Kearns G L. Proton pump inhibitors in pediatrics: mechanism of action, pharmacokinetics, pharmacogenetics, and pharmacodynamics. Pediatric Drugs. 2013, 15(2): 119-131.
PLGA NPs loaded with esomeprazole (ESO)(ESO-NPs) were developed by Cerioli and colleagues with the goal of altering the intracellular pH in a panel of different melanoma cell lines (501 mel, LM38, LM47, and LM56). ESO-NPs are taken up by melanoma cells and accumulate in melanosomal/lysosomal compartments where they release ESO, resulting in a reduction of acidity and melanin content, possibly by inhibition of tyrosinase activity. Thus, the endolysosomal compartment of cancer cells is disrupted by a pH alteration that may also affect autophagy, with possible consequences for tumor immune escape and cancer progression.
 Fig. 2 PLGA nanoparticles loaded with esomeprazole reprogram the tumor microenvironment and alleviate immunosuppression. (Cerioli N, et al. 2025)
Fig. 2 PLGA nanoparticles loaded with esomeprazole reprogram the tumor microenvironment and alleviate immunosuppression. (Cerioli N, et al. 2025)
References
- Cerioli N, et al. Reprogramming the melanoma and immunosuppressive myeloid cells with esomeprazole-loaded PLGA nanoparticles. iScience. 2025, 28(1).
What are the recommended storage temperatures for Esomeprazole?
Store Esomeprazole in a cool, dry place to maintain chemical stability.
Can I track my Esomeprazole shipment in real-time?
Yes, a tracking number is provided immediately after your Esomeprazole leaves our warehouse.
What documents accompany a shipment of Esomeprazole?
Each Esomeprazole shipment includes a COA and a detailed packing list for compliance.
What packaging is used for Esomeprazole transport?
Esomeprazole ships in moisture-proof, opaque materials.

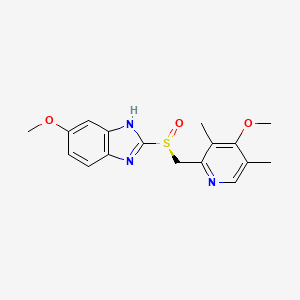
 Fig. 1 General chemical structure and mechanism of action of proton pump inhibitors (PPIs). (Ward R M, Kearns G L. 2013)
Fig. 1 General chemical structure and mechanism of action of proton pump inhibitors (PPIs). (Ward R M, Kearns G L. 2013)
 Fig. 2 PLGA nanoparticles loaded with esomeprazole reprogram the tumor microenvironment and alleviate immunosuppression. (Cerioli N, et al. 2025)
Fig. 2 PLGA nanoparticles loaded with esomeprazole reprogram the tumor microenvironment and alleviate immunosuppression. (Cerioli N, et al. 2025)













