Synonyms
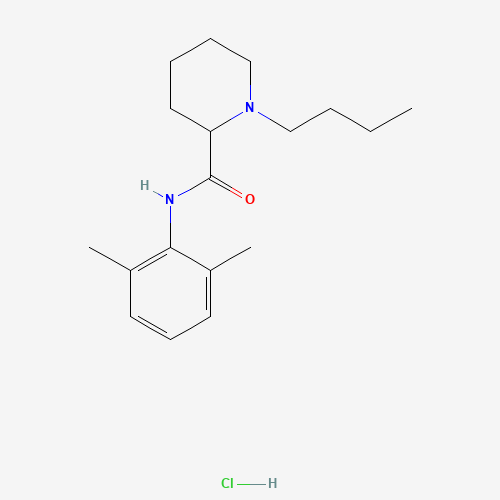
1-butyl-N-(2,6-dimethylphenyl)piperidine-2-carboxamide 38396-39-3 DL-Bupivacaine
Molecular Formula
C18H28N2O
Smiles
CCCCN1CCCCC1C(=O)NC2=C(C=CC=C2C)C
Appearance
White to off-white solid
Boiling Point
423.4±45.0℃ at 760 mmHg
General Description
Bupivacaine is a potent, long-acting local anesthetic agent belonging to the amide class. First synthesized in 1957, it is structurally related to other amides like mepivacaine and lidocaine but is distinguished by its longer duration of action and higher potency.
Mechanism of Action
The primary mechanism of action of bupivacaine, like all local anesthetics, is the reversible blockade of voltage-gated sodium channels (Nav) on neuronal cell membranes. Bupivacaine exists in a pH-dependent equilibrium between a lipophilic uncharged base and a hydrophilic charged cation. The uncharged form diffuses across the neuronal sheath and axonal membrane. Once inside the axon, it re-equilibrates, and the cationic form binds preferentially to the open/inactivated state of the sodium channel from the intracellular side . This binding physically occludes the pore, preventing sodium influx and thereby halting the propagation of the action potential.
Application
Bupivacaine is indicated for the production of local or regional anesthesia and analgesia in diagnostic and surgical procedures. Its long duration of action (2–4 hours or more, depending on dose and route) makes it a mainstay in perioperative and obstetric settings.
Chen et al. investigated the effect of bupivacaine on SK2 channels. The result showed that bupivacaine inhibited the SK2 channels reversibly in a dose-dependent manner. The IC50 value of bupivacaine on SK2 currents was 16.5µM. The degree of SK2 current inhibition by bupivacaine depended on the intracellular concentration of free calcium.
 Fig. 1 Effect of calcium concentration on the inhibitory effect of bupivacaine. (Chen H, et al., 2021)
Fig. 1 Effect of calcium concentration on the inhibitory effect of bupivacaine. (Chen H, et al., 2021)
References
- Chen H, et al. Bupivacaine inhibits a small conductance calcium-activated potassium type 2 channel in human embryonic kidney 293 cells. BMC Pharmacol Toxicol. 2021; 22(1):15.
Chen JJ, et al. conducted a meta-analysis of 16 randomized controlled trials (RCTs) revealing that patients receiving liposomal bupivacaine via periarticular injection demonstrated significantly lower morphine-equivalent opioid consumption during the critical 24- to 48-hour postoperative window compared to those receiving conventional periarticular injection.
 Fig. 2 Forest plot of morphine consumption equivalents. From top to bottom are postoperative 0–24 h, 24–48 h and 48–72 h. (Chen JJ, et al, 2023)
Fig. 2 Forest plot of morphine consumption equivalents. From top to bottom are postoperative 0–24 h, 24–48 h and 48–72 h. (Chen JJ, et al, 2023)
References
- Chen JJ, et al. Liposomal bupivacaine administration is not superior to traditional periarticular injection for postoperative pain management following total knee arthroplasty: a meta-analysis of randomized controlled trials. J Orthop Surg Res. 2023; 18(1):206.
Does Bupivacaine require protection from light during long-term storage?
Yes, it is photosensitive. Storage in light-resistant, well-closed containers is recommended to prevent photodegradation and maintain its purity.
What is the recommended storage temperature for Bupivacaine?
Store at controlled room temperature, between 15°C and 25°C. Avoid excessive heat, which can affect its crystalline structure and stability.
Is Bupivacaine stable in lipid emulsions for epidural use?
Yes, it is commonly formulated in lipid emulsions.
How is the absence of 2,6-dimethylaniline (a genotoxic impurity) verified?
We use a highly sensitive LC-MS/MS method with a detection limit well below the TTC threshold to ensure this impurity is not present in the API.


 Fig. 1 Effect of calcium concentration on the inhibitory effect of bupivacaine. (Chen H, et al., 2021)
Fig. 1 Effect of calcium concentration on the inhibitory effect of bupivacaine. (Chen H, et al., 2021)
 Fig. 2 Forest plot of morphine consumption equivalents. From top to bottom are postoperative 0–24 h, 24–48 h and 48–72 h. (Chen JJ, et al, 2023)
Fig. 2 Forest plot of morphine consumption equivalents. From top to bottom are postoperative 0–24 h, 24–48 h and 48–72 h. (Chen JJ, et al, 2023)